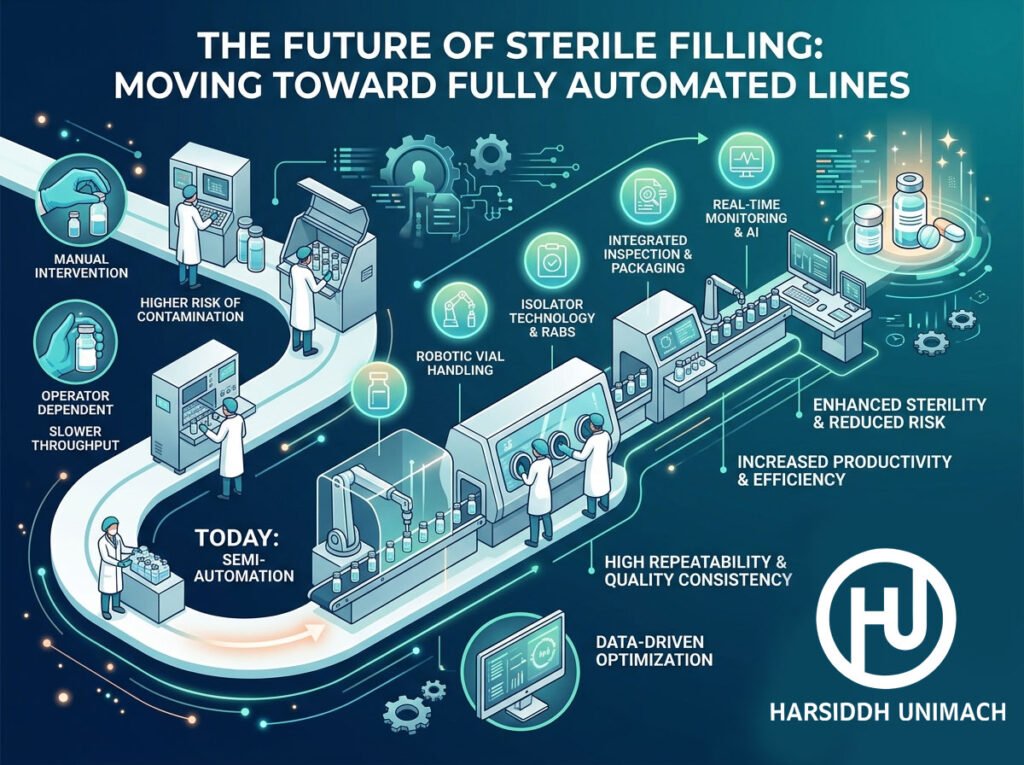
In the pharmaceutical and biotech industries, the margin for error is non-existent. As we move further into 2026, the transition from semi-automated processes to fully automated, hands-off sterile filling lines is no longer a luxury—it is a regulatory and operational necessity.
At Harsiddh Unimach, we recognize that the future of aseptic processing lies at the intersection of robotics, AI-driven monitoring, and advanced containment. Here is how the industry is evolving.
1. The Death of Human Intervention
The greatest risk to any sterile environment is, ironically, the human operator. Even with the best gowning procedures, humans are a primary source of particulate and microbial contamination.
The Shift: Modern lines are moving toward Gloveless Isolators and RABS (Restricted Access Barrier Systems). By removing the need for glove ports, manufacturers can ensure a truly hermetic seal, allowing the machine to operate in a Grade A environment without human proximity.
2. The Rise of “Batch Size One” and Flexibility
The pharmaceutical landscape is shifting from blockbuster drugs to personalized medicine and orphan drugs. This requires filling lines that can handle small batches and frequent changeovers without hours of downtime.
- Robotic Precision: Robotic arms can now swap formats (vials to syringes to cartridges) with minimal hardware changes.
- Single-Use Systems (SUS): Integrating disposable fluid paths reduces the need for complex CIP (Clean-in-Place) and SIP (Steam-in-Place) cycles, accelerating speed-to-market.
3. Real-Time Quality Control: “Zero-Loss” Filling
Traditional quality control involved sampling at the end of a run. The future is In-Process Checkweighing (IPC).
Modern lines from leaders like Harsiddh Unimach utilize 100% IPC, where every single container is weighed before and after filling. If a deviation is detected, the system adjusts the pump parameters in real-time or rejects the vial without stopping the line.
Key Formula for Filling Accuracy:
The precision of a volumetric filling system is often defined by its standard deviation. For a target volume $V_t$, the actual volume $V_a$ must satisfy:
|Vt – Va| ≤ σ
where σ is minimized through servo-driven peristaltic or rotary piston pumps.
4. Digital Twins and Predictive Maintenance
Automation isn’t just about the physical filling; it’s about the data. By creating a Digital Twin of the filling line, Harsiddh Unimach clients can simulate production runs before a single drop of product is used.
- Predictive Analytics: Instead of waiting for a motor to fail, AI sensors monitor vibration and heat signatures, alerting maintenance teams weeks before a potential breakdown occurs.
- Data Integrity: Automated lines ensure that every action is logged in compliance with 21 CFR Part 11, creating an unalterable “paperless” audit trail.
Why Harsiddh Unimach is Your Partner in This Transition
Transitioning to a fully automated line is a significant investment. At Harsiddh Unimach, we don’t just provide machines; we provide future-proofed ecosystems. Our equipment is designed with:
- Compact Footprints: Ideal for high-value cleanroom real estate.
- Modular Architecture: Allowing you to add features as your production scales.
- Global Compliance: Meeting the stringent Annex 1 requirements for aseptic processing.
The Road Ahead
The journey toward a “lights-out” manufacturing facility—where the filling process occurs entirely without human presence—is accelerating. Companies that adopt these automated technologies today will benefit from lower rejection rates, higher yield, and, most importantly, enhanced patient safety.